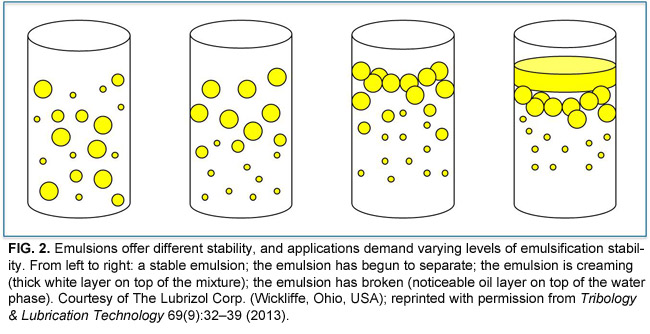
ĭestabilization of the mixture is the main purpose of the separation by separating the oil and water. It reduces the inter-facial tension between the layers of the compounds and decreases the rate of coalescence while coalescence happens when the two compound collide. Applying microwave radiation to break the emulsionĪn emulsifier’s function is to stabilize the system within a sufficient time period.Applying electrical fields that promote coalescence.Adding active chemicals compounds that are designed to the break the emulsion.To increase the probability of coalescence of dispersed water droplets on contact, the tough skin or film surrounding the dispersed water droplets must be weakened and broken. Emulsions in the oil industry are typically represented as "water-in-oil" or "oil-in-water" based on their volume of liquids ratios.Ĭounteracting the stabilizing effect of the emulsifier destabilizes an emulsion. The type of emulsions formed depends on the type of liquid which forms the the continuous phase and thus we have: (1) Oil-in-water (O/W) when oil is dispersed in water(Oil suspended in aqueous layer) and (2) water-in-oil (W/O) when water is dispersed in oil (water suspended in oil). When one liquid is dispersed in a continuous liquid phase of a different composition an emulsion is formed. By their attractive nature they lower the interfacial tension between water and oil which prevents stabilization of droplets and prevents emulsion from coalescence. Hence, they are attracted to both the polar and non-polar compounds. The emulsifier contain both hydrophilic (water loving or polar) head group and hydrophobic tail group (oil loving or non-polar). The emulsifier forms a physical layer around the compound by not allowing the droplets to coalesce. Without proper treatment, stable emulsions can take longer to separate into water and oil. Unstable emulsions can be separated into water and oil over an extended period of time. Nature, amount of emulsifying agent and degree of agitation determine the stability of the emulsions. Emulsions are formed by two immiscible liquids, emulsifying agent which is used as a stabilizer and agitation for converting from discontinuous phase to continuous phase. 
Separation requires factors other than gravity. A mixture of oil and water which is stable and cannot be separated by gravity settling alone.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
