|
The ground state of the nitrogen atom is a 4S state, for which 2S + 1 = 4 in a quartet state, S = 3/2 due to three unpaired electrons. When S > L there are only 2L+1 orientations of total angular momentum possible, ranging from S+L to S-L. 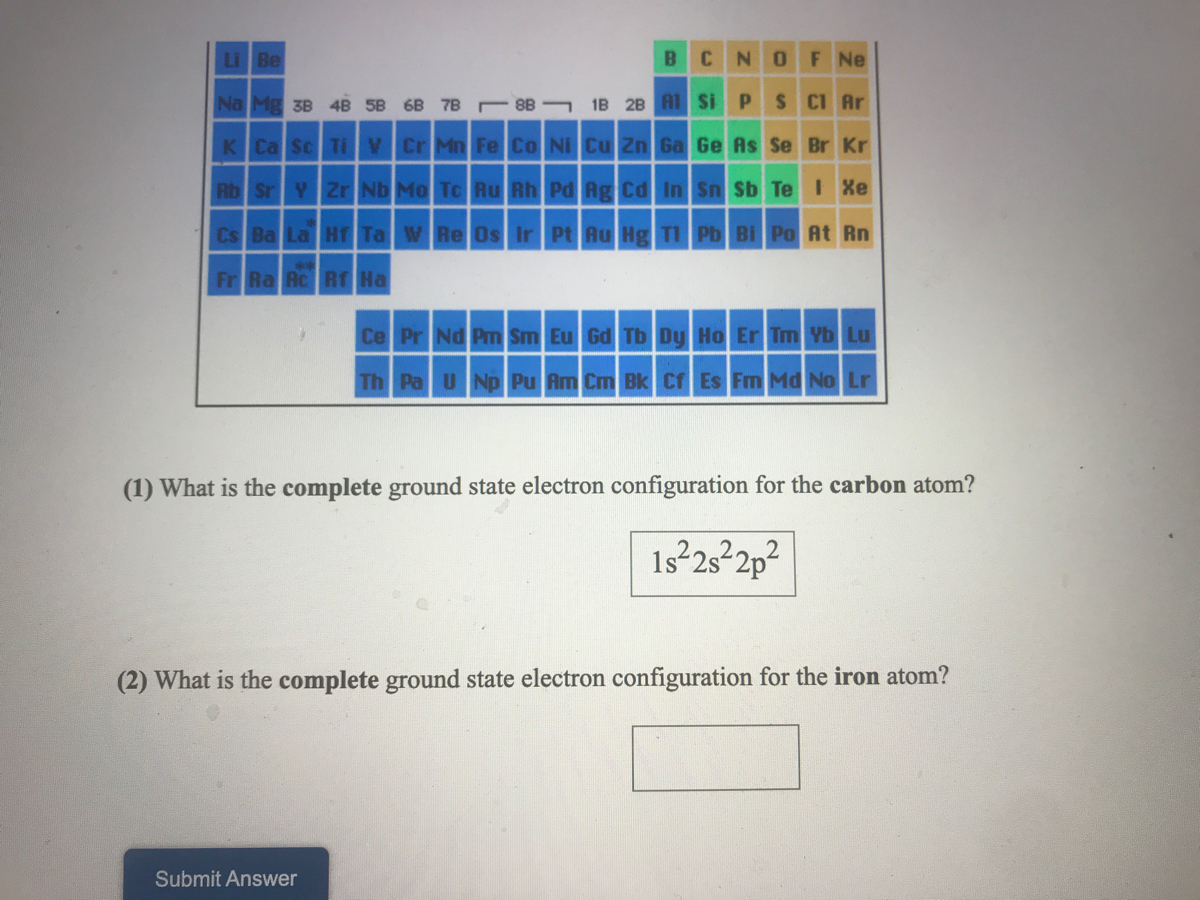
However the multiplicity equals the number of spin orientations only if S ≤ L. The total angular momentum quantum number J can vary from L+S = 2 to L–S = 0 in integer steps, so that J = 2, 1 or 0. The triplet consists of three states with spin components +1, 0 and –1 along the direction of the total orbital angular momentum, which is also 1 as indicated by the letter P. This spin is due to two unpaired electrons, as a result of Hund's rule which favors the single filling of degenerate orbitals. The superscript three (read as triplet) indicates that the multiplicity 2S+1 = 3, so that the total spin S = 1. The multiplicity is often equal to the number of possible orientations of the total spin relative to the total orbital angular momentum L, and therefore to the number of near– degenerate levels that differ only in their spin–orbit interaction energy.įor example, the ground state of a carbon atom is 3P. In this case the multiplicity is also equal to the number of unpaired electrons plus one. In the ground state of an atom or molecule, the unpaired electrons usually all have parallel spin. States with multiplicity 1, 2, 3, 4, 5 are respectively called singlets, doublets, triplets, quartets and quintets. In spectroscopy and quantum chemistry, the multiplicity of an energy level is defined as 2S+1, where S is the 
Property of an atomic or molecular energy level
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
